Pharmacological treatment of polycystic ovary syndrome – health and quality of life in the short and long term
A systematic review including health economic and ethical aspects
Main message
Polycystic ovary syndrome (PCOS) is a common condition in women, caused by hormonal variations. Pharmacological treatments for PCOS can have some positive short-term effects, but there are major evidence gaps about their long-term effectiveness and safety. This report investigates the effects of combined oral contraceptive pills (COCPs), antiandrogen drugs, metformin and glucagon-like peptide-1 analogues (GLP-1 analogues) in individuals with PCOS.
Conclusions
- COCPs and metformin may improve menstrual frequency and regularity. The evidence for GLP-1 analogues and antiandrogens is insufficient to assess their effect on menstruation.
- Hirsutism, excessive growth of hair on the body or face, is a common symptom in women with PCOS. COCPs that contain cyproterone acetate (CPA) may have a slightly better effect on hirsutism compared to regular COCPs. For other comparisons between types of COCPs, or COCPs vs. no treatment, evidence is insufficient. Antiandrogens, whether alone or as add-ons, do not seem effective, but the evidence base is limited.
- Metformin and GLP-1 analogues can lead to some weight loss in individuals with overweight or obesity. Evidence is insufficient for weight effects of COCPs and antiandrogens. Metformin provides modest improvement in markers for glucose and insulin regulation but shows no effect on blood lipids.
- For all the evaluated drugs, the evidence is insufficient to assess their effects on quality of life.
- There is also insufficient evidence to determine the long-term health effects of these treatments for PCOS. This is due to a lack of long-term studies.
Aim
The purpose of this systematic review was to evaluate the efficacy, safety, short and long-term health impacts, health-economic aspects, and ethical considerations of pharmacological treatments currently used – or potentially applicable – for individuals diagnosed with PCOS.
Background
PCOS is a common hormonal disorder in women of reproductive age, affecting about 12% of adult women. It is characterized by elevated androgen levels, irregular or absent menstruation, and multiple ovarian follicles. PCOS is also associated with insulin resistance, overweight or obesity, infertility, and increased risk for type 2 diabetes, cardiovascular disease, endometrial cancer, and mental health issues such as anxiety and depression.
Diagnosis typically requires at least two out of three criteria: hyperandrogenism, ovulatory dysfunction, and polycystic ovarian morphology. The condition presents differently across individuals and over the lifespan.
There is no cure for PCOS; treatments aim to manage symptoms. Lifestyle changes are first-line therapy, especially for individuals with overweight or obesity. Pharmacological treatments – often off-label – include COCPs, metformin and antiandrogens. GLP-1 analogues can be used in patients with type 2 diabetes or obesity.
The healthcare system often addresses symptoms in isolation, which may lead to fragmented care. A holistic, multidisciplinary approach is increasingly recommended. Updated international guidelines were released in 2023, but Sweden lacks a national care program for PCOS, motivating this comprehensive evidence review.
Method
We conducted a systematic review following the standard methods used by SBU and reported it in accordance with the PRISMA statement. The protocol was registered in Prospero (CRD42024535096). The review included randomized controlled trials (RCTs) and observational studies (for long-term outcomes), used meta-analyses when possible, and applied the GRADE system to assess certainty of evidence. Additional literature searches were conducted for health economic studies and a chapter on ethical aspects was included.
Inclusion criteria
- Population: Women of all ages with PCOS diagnosed per Rotterdam, NIH, or AES criteria. Studies on pregnant women were excluded or if pregnant patients were included, they had to be reported separately. In register-based studies diagnostic code equivalent with a clinical diagnosis of PCOS was required.
- Intervention: COCPs, antiandrogen drugs, metformin, GLP-1 analogues.
- Control: Placebo, no treatment, lifestyle intervention, or comparison with other drug/drugs in one or several of the intervention categories.
- Outcomes: Menstrual regularity, hirsutism, body mass index (BMI), waist-hip ratio (WHR), insulin/glucose markers (fasting insulin, fasting glucose and HOMA-IR), blood lipids (triglycerides (TG), LDL (low density lipoprotein) cholesterol, psychological outcomes, quality of life, adverse effects, long-term outcomes (e.g. diabetes, CVD, cancer, mortality).
- Study design: RCTs for short-term effects; RCTs and cohort/case-control studies (≥100 participants) for long-term outcomes.
- Language: English, Swedish, Norwegian, or Danish.
Databases searched: MEDLINE (Ovid), EMBASE, Cochrane Library, CINAHL, PsycINFO. Last literature search was performed in Oct 2024. - Health economic assessment: for GLP-1-analogues the literature was screened for health economic assessments for cost and cost-effectiveness.
- Patient involvement: Input from a patient representative helped shape outcome priorities and ethical discussions.
Results
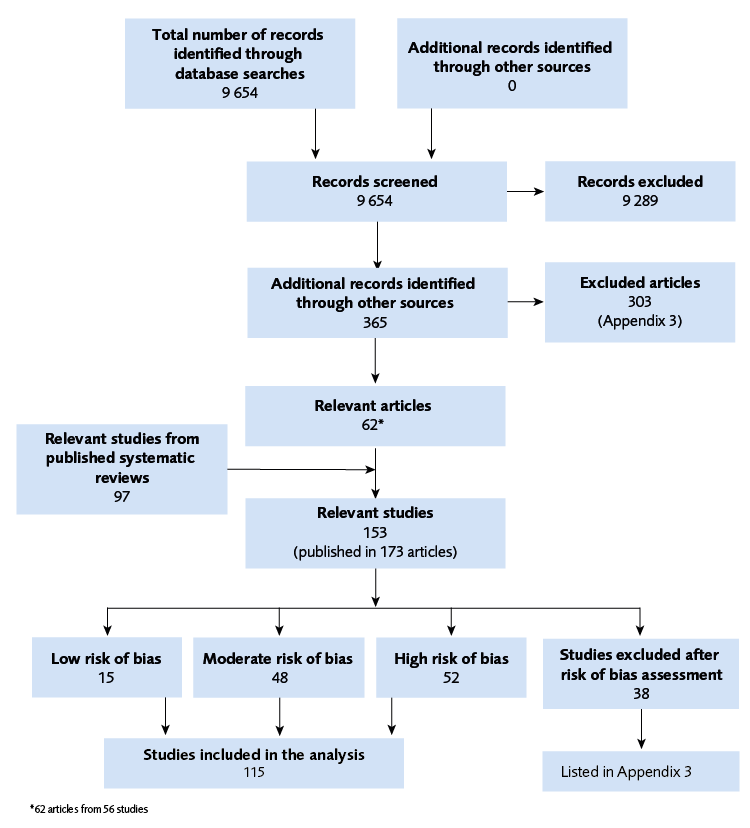
A total of 9654 abstracts were identified by the literature search, 365 were read in full text and 62 articles (56 studies) were eligible. To these another 97 eligible studies were added from five previously published systematic reviews, yielding a total of 153 relevant studies. A further 38 were excluded as they were determined to have a critical risk of bias, leaving 115 studies included in the analyses, see Figure 1.
Combined oral contraceptive pills
When COCPs were compared to no treatment, menstrual regularity and frequency was improved, with low certainty (⊕⊕◯◯). For all other outcomes the level of certainty was very low (⊕◯◯◯). No studies on this comparison were added in the updated search for this report.
In the comparison between different types of COCPs, one study was added in this report, compared to the previously published systematic review. Most comparisons had a result with very low certainty of evidence (⊕◯◯◯). However, patients treated with contraceptive pills containing cyproterone acetate and ethyl estradiol had a lower BMI and reduced levels of hirsutism after treatment compared to patients treated with other types of COCPs, both results with low certainty (⊕⊕◯◯). For fasting insulin and the same comparison, no difference was observed (⊕⊕◯◯).
Antiandrogen drugs
In the comparison of different types of antiandrogen drugs and other treatments or no treatment, there was no added effect of antiandrogen drugs regarding hirsutism, fasting glucose or triglycerides, with low certainty of evidence (⊕⊕◯◯). For all other outcomes the level of certainty was very low (⊕◯◯◯). No studies were added in the updated search for this report, but we performed additional meta-analyses.
Metformin and combined oral contraceptive pills
In comparisons between COCPs alone and in combination with metformin, no differences were found in BMI (⊕⊕◯◯), WHR (⊕⊕⊕◯), or hirsutism (⊕⊕⊕◯). Fasting insulin and HOMA-IR improved slightly more with combination treatment (⊕⊕◯◯) while fasting glucose showed no difference (⊕⊕◯◯). Lipid levels also showed no differences between groups (low to moderate certainty).
When metformin plus COCPs were compared to metformin alone, most of our prioritized outcomes had very low certainty (⊕◯◯◯). No differences were found for fasting insulin or LDL cholesterol (⊕⊕◯◯ ).
No studies were added in the updated search for this report.
Metformin
Three studies were found in the updated search for this report and added to the 29 studies included in the original systematic review. New meta-analyses were performed comparing studies were metformin with or without lifestyle interventions were compared to no treatment, placebo or lifestyle interventions only.
| Outcome | Meta-analysis (patients; studies) * |
Results (favors; point estimate; 95% confidence interval) |
GRADE |
|---|---|---|---|
| BMI | 312 patients 6 studies |
Favors GLP-1 analogues –1.38 (–2.39 to –0.38) |
⊕⊕◯◯ |
| WHR | 192 patients 3 studies |
No difference –0.01 (–0.10 to 0.08) |
⊕◯◯◯ |
| Fasting glucose | 319 patients 5 studies |
No difference –0.10 (–0.46 to 0.26) |
⊕◯◯◯ |
| Fasting insulin | 192 patients 3 studies |
No difference 9.72 (–30.14 to 49.59) |
⊕◯◯◯ |
| HOMA-IR | 260 patients 4 studies |
No difference –0.28 (–2.36 to 1.80) |
⊕◯◯◯ |
| LDL cholesterol | 260 patients 5 studies |
No difference 0.01 (–0.04 to 0.05) |
⊕⊕⊕◯ |
| Triglycerides | 127 patients 2 studies |
No difference –0.05 (–0.26 to 0.17) |
⊕⊕◯◯ |
| Hirsutism | 72 patients 1 study** |
No difference |
⊕◯◯◯ |
| Menstruation | 250 patients 5 studies |
Favors GLP-1 analogues (synthesis without meta-analysis) |
⊕◯◯◯ |
| Abbreviations: BMI= body mass index; GLP-1 analogues= glucagone like peptide 1 analogues; LDL= low density lipoprotein; WHR= waist-hip-ratio *In the original report, studies that reported results that could not be included in a meta-analysis were reported narratively and reported in the summary of findings table ** Result in study only reported narratively |
|||
Long-term outcomes of pharmacological treatments
The literature search identified five register-based studies that filled the inclusion criteria. Also, twenty randomized controlled studies on COCPs, antiandrogen drugs or metformin had a follow up period of 12 months or longer. No long-term studies on the effects or side effects of GLP-1-analogues were identified.
The scientific evidence was insufficient to assess the impact of these pharmacological treatments on important health outcomes for patients with PCOS, such as diabetes, cardiovascular disease, mental health disorders, or endometrial changes.
Treatment with COCPs containing CPA likely improves hirsutism more effectively than standard combined contraceptives after 12 months (⊕⊕◯◯). However, this benefit must be weighed against the known risks associated with CPA. For all other outcomes assessed in RCTs, such as menstrual regularity, hirsutism, weight related or metabolic outcomes, no conclusions could be drawn regarding long-term health and treatment with COCPs, antiandrogen drugs and metformin.
Health Economic Assessment
The health economic review focused on GLP-1 analogues due to their higher cost level compared to the other pharmacological treatments evaluated in this report. No cost-effectiveness studies specific to PCOS were found for GLP-1 analogues. The assessment also mapped Swedish reimbursement status for all the drugs included in this report.
Ethical and societal aspects
PCOS is a common condition were patients in Sweden have unequal access to care across regions and care levels, often leading to fragmented treatment. Many medications are used off-label without strong evidence, raising ethical concerns, especially regarding the long-term benefit of treatments. There may be stigma associated to certain aspects of PCOS, such as hirsutism and obesity, which can complicate care. Disparities in access to costly treatments like GLP-1-analogues may worsen health inequality. There is also concern about healthcare system strain and treatment crowd-out. Given several knowledge gaps, treatment decisions must be individualized, transparent about uncertainties and based on both professional judgment and patient preferences.
Discussion
This report shows that several pharmacological treatments can relieve important symptoms for patients with PCOS, such as irregular menstruation and excess weight. We conclude that metformin could have a small effect on weight loss, however the effect of metformin on metabolic outcomes is modest. GLP-1 analogues may support short-term weight loss but lack solid evidence for broader benefits or safety. As of hirsutism, only CPA-containing pills have some evidence for reducing that outcome, though risks of adverse events limit their use.
COCP will likely improve menstrual regularity in PCOS women in the same manner that is observed in healthy women. It is however not studied what effect a continuous use of COCPs (no pill breaks) would have on the endothelium in women with PCOS.
When assessing the research for all included interventions, we concluded that research is often limited to short follow-up periods, surrogate outcomes, and a narrow age range of study-participants. This means that the long-term health effects for all investigated pharmacological treatments remain unclear.
This is an evidence gap of significance and longer studies are needed in order to investigate if pharmacological treatments can reduce the risk of co-morbidities such as diabetes type 2, cardiovascular disease and endometrial cancer in women with PCOS.
Also, most included studies did not assess quality of life or investigated the effects on mental health. It is important that these outcomes are included in future studies.
Conflict of Interest
The experts and scientific reviewers participating in this project have submitted statements about conflicts of interest. These documents are available at SBU’s secretariat. SBU has determined that the conditions described in the submissions are compatible with SBU’s requirements for objectivity and impartiality.
The full report in Swedish
Läkemedelsbehandling av polycystiskt ovarialsyndrom – hälsa och livskvalitet på kort och lång sikt
Project group
Experts
- Angelica Lindén Hirschberg, Professor, Department of Women’s and Children’s Health, Karolinska Institutet; Senior Physician, Gynecology and Obstetrics, Karolinska University Hospital
- Maria Forslund, Associate Professor, Sahlgrenska Academy, University of Gothenburg; Senior Physician, Gynecology and Obstetrics, Sahlgrenska University Hospital
- Per Wändell, Professor Emeritus in Family Medicine, Department of Neurobiology, Care Sciences and Society, Karolinska Institutet
SBU
- Lisa Forsberg, Project Director
- Marie Österberg, Assistant Project Director
- Jessica Dagerhamn, Assistant Project Director
- Jenny Ågren, Analyst
- Maja Kärrman Fredriksson, Information Specialist
- Emma Wernersson, Project Administrator
- Sigrid Widén, Project Administrator
- Maria Hoppe, Project Administrator
- Anna Ringborg, Health Economist
- Jenny Odeberg, Head of department
Flow chart
References
- Teede HJ, Tay CT, Laven JJE, Dokras A, Moran LJ, Piltonen TT, et al. Recommendations from the 2023 international evidence-based guideline for the assessment and management of polycystic ovary syndrome. European Journal of Endocrinology. 2023;189(2):G43-G64. Available from: https://doi.org/10.1093/ejendo/lvad096
- Spritzer PM, Marchesan LB, Santos BR, Fighera TM. Hirsutism, Normal Androgens and Diagnosis of PCOS. Diagnostics (Basel). 2022;12(8). Available from: https://doi.org/10.3390/diagnostics12081922
- Liu X, Zhang J, Wang S. Global, regional, and national burden of infertility attributable to PCOS, 1990–2019. Human Reproduction. 2023;39(1):108-18. Available from: https://doi.org/10.1093/humrep/dead241
- Li Z, Wang YH, Wang LL, Hu DT, Teng Y, Zhang TY, et al. Polycystic ovary syndrome and the risk of endometrial, ovarian and breast cancer: An updated meta-analysis. Scott Med J. 2022;67(3):109-20. Available from: https://doi.org/10.1177/00369330221107099
- Forslund M, Melin J, Stener-Victorin E, Hirschberg AL, Teede H, Vanky E, et al. International evidence-based guideline on assessment and management of PCOS-A Nordic perspective. Acta Obstet Gynecol Scand. 2024;103(1):7-12. Available from: https://doi.org/10.1111/aogs.14725
- Kakoly NS, Khomami MB, Joham AE, Cooray SD, Misso ML, Norman RJ, et al. Ethnicity, obesity and the prevalence of impaired glucose tolerance and type 2 diabetes in PCOS: a systematic review and meta-regression. Hum Reprod Update. 2018;24(4):455-67. Available from: https://doi.org/10.1093/humupd/dmy007
- Glintborg D, Ollila MM, Møller JK, Pesonen P, Persson S, Elenis E, et al. Prospective risk of Type 2 diabetes in 99 892 Nordic women with polycystic ovary syndrome and 446 055 controls: national cohort study from Denmark, Finland, and Sweden. Hum Reprod. 2024;39(8):1823-34. Available from: https://doi.org/10.1093/humrep/deae124
- Lim SS, Kakoly NS, Tan JWJ, Fitzgerald G, Bahri Khomami M, Joham AE, et al. Metabolic syndrome in polycystic ovary syndrome: a systematic review, meta-analysis and meta-regression. Obes Rev. 2019;20(2):339-52. Available from: https://doi.org/10.1111/obr.12762
- Oberg E, Lundell C, Blomberg L, Gidlöf SB, Egnell PT, Hirschberg AL. Psychological well-being and personality in relation to weight loss following behavioral modification intervention in obese women with polycystic ovary syndrome: a randomized controlled trial. European Journal of Endocrinology. 2020;183(1):1-11. Available from: https://doi.org/10.1530/eje-20-0066
- Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81(1):19-25. Available from: https://doi.org/10.1016/j.fertnstert.2003.10.004
- Neven ACH, Forslund M, Ranashinha S, Mousa A, Tay CT, Peña A, et al. Prevalence and accurate diagnosis of polycystic ovary syndrome in adolescents across world regions: a systematic review and meta-analysis. Eur J Endocrinol. 2024;191(4):S15-s27. Available from: https://doi.org/10.1093/ejendo/lvae125
- Piltonen TT, Viita-Aho J, Saarela U, Melin J, Forslund M. Utility of Serum Anti-Müllerian Hormone Measurement as Part of Polycystic Ovary Syndrome Diagnosis. Semin Reprod Med. 2024;42(1):49-59. Available from: https://doi.org/10.1055/s-0044-1786731
- Mumusoglu S, Yildiz BO. Polycystic ovary syndrome phenotypes and prevalence: Differential impact of diagnostic criteria and clinical versus unselected population. Current Opinion in Endocrine and Metabolic Research. 2020;12:66-71. Available from: https://doi.org/10.1016/j.coemr.2020.03.004
- Joham AE, Norman RJ, Stener-Victorin E, Legro RS, Franks S, Moran LJ, et al. Polycystic ovary syndrome. Lancet Diabetes Endocrinol. 2022;10(9):668-80. Available from: https://doi.org/10.1016/s2213-8587(22)00163-2
- Escobar-Morreale HF. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nature Reviews Endocrinology. 2018;14(5):270-84. Available from: https://doi.org/10.1038/nrendo.2018.24
- Simon SL, Phimphasone-Brady P, McKenney KM, Gulley LD, Bonny AE, Moore JM, et al. Comprehensive transition of care for polycystic ovary syndrome from adolescence to adulthood. Lancet Child Adolesc Health. 2024;8(6):443-55. Available from: https://doi.org/10.1016/s2352-4642(24)00019-1
- Bahri Khomami M, Hashemi S, Shorakae S, Harrison CL, Piltonen TT, Romualdi D, et al. Systematic review and meta-analysis of birth outcomes in women with polycystic ovary syndrome. Nature Communications. 2024;15(1):5592. Available from: https://doi.org/10.1038/s41467-024-49752-6
- Forslund M, Schmidt J, Brännström M, Landin-Wilhelmsen K, Dahlgren E. Reproductive Hormones and Anthropometry: A Follow-Up of PCOS and Controls From Perimenopause to Older Than 80 Years. J Clin Endocrinol Metab. 2021;106(2):421-30. Available from: https://doi.org/10.1210/clinem/dgaa840
- Millán-de-Meer M, Luque-Ramírez M, Nattero-Chávez L, Escobar-Morreale HF. PCOS during the menopausal transition and after menopause: a systematic review and meta-analysis. Hum Reprod Update. 2023;29(6):741-72. Available from: https://doi.org/10.1093/humupd/dmad015
- Tay CT, Mousa A, Vyas A, Pattuwage L, Tehrani FR, Teede H. 2023 International Evidence-Based Polycystic Ovary Syndrome Guideline Update: Insights From a Systematic Review and Meta-Analysis on Elevated Clinical Cardiovascular Disease in Polycystic Ovary Syndrome. J Am Heart Assoc. 2024;13(16):e033572. Available from: https://doi.org/10.1161/jaha.123.033572
- Hirschberg AL. Approach to Investigation of Hyperandrogenism in a Postmenopausal Woman. The Journal of Clinical Endocrinology & Metabolism. 2022;108(5):1243-53. Available from: https://doi.org/10.1210/clinem/dgac673
- Kiconco S, Tay CT, Rassie KL, Azziz R, Teede HJ, Joham AE. Natural history of polycystic ovary syndrome: A systematic review of cardiometabolic outcomes from longitudinal cohort studies. Clin Endocrinol (Oxf). 2022;96(4):475-98. Available from: https://doi.org/10.1111/cen.14647
- MCHRI. International Evidence-based Guideline for the assessment and management of polycystic ovary syndrome 2023. Melbourne: Monash Centre for Health Research and Implementation (MCHRI); 2023. [accessed Jul 8 2025]. Available from: https://www.monash.edu/__data/assets/pdf_file/0003/3379521/Evidence-Based-Guidelines-2023.pdf
- Forslund M, Melin J, Alesi S, Piltonen T, Romualdi D, Tay CT, et al. Different kinds of oral contraceptive pills in polycystic ovary syndrome: a systematic review and meta-analysis. European Journal of Endocrinology. 2023;189(1):S1-S16. Available from: https://doi.org/10.1093/ejendo/lvad082
- Hälso-och sjukvårdsförvaltningen. Diabetes mellitus typ 2. 2025. [updated 28 Mar 2025]. Available from: https://klokalistan.se/terapiomrade/endokrinologi/diabetes-mellitus-typ-2.html
- Roberts C-T, Raabe N, Wiegand L, Kadar Shahib A, Rastegar M. Diverse Applications of the Anti-Diabetic Drug Metformin in Treating Human Disease. Pharmaceuticals. 2024;17(12):1601.
- Teede HJ, Misso ML, Costello MF, Dokras A, Laven J, Moran L, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril. 2018;110(3):364-79. Available from: https://doi.org/10.1016/j.fertnstert.2018.05.004
- Teede HJ, Gibson M, Laven J, Dokras A, Moran LJ, Piltonin T, et al. International PCOS guideline clinical research priorities roadmap: a co-designed approach aligned with end-user priorities in a neglected women's health condition. EClinicalMedicine. 2024;78:102927. Available from: https://doi.org/10.1016/j.eclinm.2024.102927
- SBU. Utvärdering av metoder i hälso- och sjukvården och insatser i socialtjänsten: en metodbok. Stockholm: Statens beredning för medicinsk och social utvärdering (SBU); 2024. Available from: https://www.sbu.se/sv/metod/metodboken-2023/
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. Available from: https://doi.org/10.1371/journal.pmed.1000097
- PROSPERO. National Institute for Health and Care Research. [accessed Jul 11 2025]. Available from: https://www.crd.york.ac.uk/PROSPERO/login
- Al Wattar BH, Teede H, Garad R, Franks S, Balen A, Bhide P, et al. Harmonising research outcomes for polycystic ovary syndrome: an international multi-stakeholder core outcome set. Human Reproduction. 2020;35(2):404-12. Available from: https://doi.org/10.1093/humrep/dez272
- Forslund M, Melin J, Alesi S, Piltonen T, Romualdi D, Tay CT, et al. Combined oral contraceptive pill compared with no medical treatment in the management of polycystic ovary syndrome: A systematic review. Clinical Endocrinology. 2023;99(1):79-91. Available from: https://doi.org/10.1111/cen.14913
- Melin J, Forslund M, Alesi S, Piltonen T, Romualdi D, Spritzer PM, et al. Metformin and Combined Oral Contraceptive Pills in the Management of Polycystic Ovary Syndrome: A Systematic Review and Meta-analysis. The Journal of Clinical Endocrinology & Metabolism. 2023;109(2):e817-e36. Available from: https://doi.org/10.1210/clinem/dgad465
- Melin J, Forslund M, Alesi S, Piltonen T, Romualdi D, Spritzer PM, et al. The impact of metformin with or without lifestyle modification versus placebo on polycystic ovary syndrome: a systematic review and meta-analysis of randomized controlled trials. European Journal of Endocrinology. 2023;189(2):S38-S64. Available from: https://doi.org/10.1093/ejendo/lvad098
- Alesi S, Forslund M, Melin J, Romualdi D, Peña A, Tay CT, et al. Efficacy and safety of anti-androgens in the management of polycystic ovary syndrome: a systematic review and meta-analysis of randomised controlled trials. eClinicalMedicine. 2023;63:102162. Available from: https://doi.org/10.1016/j.eclinm.2023.102162
- Ladson G, Dodson WC, Sweet SD, Archibong AE, Kunselman AR, Demers LM, et al. The effects of metformin with lifestyle therapy in polycystic ovary syndrome: a randomized double-blind study. Fertil Steril. 2011;95(3):1059-66.e1-7. Available from: https://doi.org/10.1016/j.fertnstert.2010.12.002
- Onalan G, Goktolga U, Ceyhan T, Bagis T, Onalan R, Pabuçcu R. Predictive value of glucose-insulin ratio in PCOS and profile of women who will benefit from metformin therapy: obese, lean, hyper or normoinsulinemic? Eur J Obstet Gynecol Reprod Biol. 2005;123(2):204-11. Available from: https://doi.org/10.1016/j.ejogrb.2005.05.010
- SBU. Bedömning av systematiska översikter (ROBIS). Stockholm: Statens beredning för medicinsk och social utvärdering; 2020. [accessed Feb 29 2024]. Available from: https://www.sbu.se/globalassets/ebm/bedomning_systematiska_oversikter_robis.pdf
- Deeks JJ, Higgins JP, Altman DG, McKenzie JE, Veroniki AA. Chapter 10: Chapter 10.5.2: Meta-analysis of change scores [last updated November 2024]. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al., editors. Cochrane Handbook för Systematic Reviews of Interventions version 65: Cochrane; 2024.
- Lingaiah S, Morin-Papunen L, Risteli J, Tapanainen JS. Metformin decreases bone turnover markers in polycystic ovary syndrome: a post hoc study. Fertility and Sterility. 2019;112(2):362-70. Available from: https://doi.org/10.1016/j.fertnstert.2019.04.013
- Morin-Papunen L, Rantala AS, Unkila-Kallio L, Tiitinen A, Hippelainen M, Perheentupa A, et al. Metformin improves pregnancy and live-birth rates in women with polycystic ovary syndrome (PCOS): a multicenter, double-blind, placebo-controlled randomized trial. J Clin Endocrinol Metab. 2012;97(5):1492-500. Available from: https://doi.org/10.1210/jc.2011-3061
- Covidence. Covidence systematic review software. Melbourne, Australia: Veritas Health Innovation. Available from: Covidence - Better systematic review management
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. Bmj. 2019;366:l4898. Available from: https://doi.org/10.1136/bmj.l4898
- Sterne JA, Hernan MA, Reeves BC, Savovic J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. Available from: https://doi.org/10.1136/bmj.i4919
- SBU. Manual till mallarna för randomiserade och icke randomiserade interventionsstudier. Stockholm: Statens beredning för medicinsk och social utvärdering (SBU); 2022. [updated May 11 2022]. Available from: https://www.sbu.se/globalassets/ebm/manual_mallarna_randomiserade_icke-randomiserade_kontrollerade_studier.pdf
- Goldberg A, Graca S, Liu J, Rao V, Witchel SF, Pena A, et al. Anti-obesity pharmacological agents for polycystic ovary syndrome: A systematic review and meta-analysis to inform the 2023 international evidence-based guideline. Obes Rev. 2024;25(5):e13704. Available from: https://doi.org/10.1111/obr.13704
- The Nordic Cochrane Centre TCC. Review Manager (RevMan). Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration. Available from: https://www.cochrane.org/learn/courses-and-resources/software
- Higgins J, Thomas J, Chandler J, Cumpston M, Li T, Page M, et al. Cochrane Handbook for Systematic Reviews of Interventions version 6.5 (updated August 2024). Cochrane; 2024. Available from: www.training.cochrane.org/handbook
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction - GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology. 2011;64(4):383-94. Available from: https://doi.org/10.1016/j.jclinepi.2010.04.026
- Schünemann HB, J., Guyatt G, Oxman A, editors. GRADE handbook for grading quality of evidence and strength of recommendations; 2013. [accessed Feb 29 2024]. Available from: https://gdt.gradepro.org/app/handbook/handbook.html
- SBU. Etiska aspekter på insatser inom hälso- och sjukvården. En vägledning för att identifiera relevanta etiska aspekter. Stockholm: Statens beredning för medicinsk och social utvärdering (SBU); 2021. Available from: https://www.sbu.se/globalassets/ebm/etiska_aspekter_halso_sjukvarden.pdf
- Jahanfar S, Mortazavi J, Lapidow A, Cu C, Al Abosy J, Ciana H, et al. Assessing the impact of hormonal contraceptive use on menstrual health among women of reproductive age - a systematic review. Eur J Contracept Reprod Health Care. 2024;29(5):193-223. Available from: https://doi.org/10.1080/13625187.2024.2373143
- Dasgupta S, Mondal J, Goswami B, Dasgupta J. Randomized control trial to compare effects of ultra-low dose (Ethinylestradiol 20 μg or 15 μg) with low dose (Ethinylestradiol 30 μg) hormonal pills on lipid discordance in non-obese PCOS women. Obstet Gynecol Sci. 2023;66(6):572-83. Available from: https://doi.org/10.5468/ogs.23142
- Falsetti L, Gambera A, Legrenzi L, Iacobello C, Bugari G. RETRACTED: Comparison of finasteride versus flutamide in the treatment of hirsutism. European Journal of Endocrinology. 1999;141(4):361-7. Available from: https://doi.org/10.1530/eje.0.1410361
- Amiri M, Golsorkhtabaramiri M, Esmaeilzadeh S, Ghofrani F, Bijani A, Ghorbani L, et al. Effect of Metformin and Flutamide on Anthropometric Indices and Laboratory Tests in Obese/Overweight PCOS Women under Hypocaloric Diet. J Reprod Infertil. 2014;15(4):205-13.
- Diri H, Bayram F, Simsek Y, Caliskan Z, Kocer D. COMPARISON OF FINASTERIDE, METFORMIN, AND FINASTERIDE PLUS METFORMIN IN PCOS. Acta Endocrinol (Buchar). 2017;13(1):84-9. Available from: https://doi.org/10.4183/aeb.2017.84
- Dumesic DA, Winnett C, Lu G, Grogan TR, Abbott DH, Naik R, et al. Randomized clinical trial: effect of low-dose flutamide on abdominal adipogenic function in normal-weight women with polycystic ovary syndrome. Fertil Steril. 2023;119(1):116-26. Available from: https://doi.org/10.1016/j.fertnstert.2022.09.324
- Gambineri A, Patton L, Vaccina A, Cacciari M, Morselli-Labate AM, Cavazza C, et al. Treatment with flutamide, metformin, and their combination added to a hypocaloric diet in overweight-obese women with polycystic ovary syndrome: a randomized, 12-month, placebo-controlled study. J Clin Endocrinol Metab. 2006;91(10):3970-80. Available from: https://doi.org/10.1210/jc.2005-2250
- Ganie MA, Khurana ML, Nisar S, Shah PA, Shah ZA, Kulshrestha B, et al. Improved efficacy of low-dose spironolactone and metformin combination than either drug alone in the management of women with polycystic ovary syndrome (PCOS): a six-month, open-label randomized study. J Clin Endocrinol Metab. 2013;98(9):3599-607. Available from: https://doi.org/10.1210/jc.2013-1040
- Mazza A, Fruci B, Guzzi P, D'Orrico B, Malaguarnera R, Veltri P, et al. In PCOS patients the addition of low-dose spironolactone induces a more marked reduction of clinical and biochemical hyperandrogenism than metformin alone. Nutrition, Metabolism and Cardiovascular Diseases. 2014;24(2):132-9. Available from: https://doi.org/10.1016/j.numecd.2013.04.016
- Vieira CS, Martins WP, Fernandes JB, Soares GM, dos Reis RM, de Sá MF, et al. The effects of 2 mg chlormadinone acetate/30 mcg ethinylestradiol, alone or combined with spironolactone, on cardiovascular risk markers in women with polycystic ovary syndrome. Contraception. 2012;86(3):268-75. Available from: https://doi.org/10.1016/j.contraception.2011.12.011
- Wen Q, Hu M, Lai M, Li J, Hu Z, Quan K, et al. Effect of acupuncture and metformin on insulin sensitivity in women with polycystic ovary syndrome and insulin resistance: a three-armed randomized controlled trial. Human Reproduction. 2022;37(3):542-52.
- Cao J, Nie G, Dai Z, Shan D, Wei Z. Comparative effects of acupuncture and metformin on insulin sensitivity in overweight/obese and lean women with polycystic ovary syndrome and insulin resistance: a post hoc analysis of a randomized trial. Front Med (Lausanne). 2023;10:1232127. Available from: https://doi.org/10.3389/fmed.2023.1232127
- Dilimulati D, Shao X, Wang L, Cai M, Zhang Y, Lu J, et al. Efficacy of WeChat-Based Digital Intervention Versus Metformin in Women With Polycystic Ovary Syndrome: Randomized Controlled Trial. Journal of Medical Internet Research. 2024;26:e55883.
- Hoeger K, Davidson K, Kochman L, Cherry T, Kopin L, Guzick DS. The impact of metformin, oral contraceptives, and lifestyle modification on polycystic ovary syndrome in obese adolescent women in two randomized, placebo-controlled clinical trials. J Clin Endocrinol Metab. 2008;93(11):4299-306. Available from: https://doi.org/10.1210/jc.2008-0461
- Esfahanian F, Zamani MM, Heshmat R, Moini nia F. Effect of metformin compared with hypocaloric diet on serum C-reactive protein level and insulin resistance in obese and overweight women with polycystic ovary syndrome. J Obstet Gynaecol Res. 2013;39(4):806-13. Available from: https://doi.org/10.1111/j.1447-0756.2012.02051.x
- Elkind-Hirsch K, Marrioneaux O, Bhushan M, Vernor D, Bhushan R. Comparison of single and combined treatment with exenatide and metformin on menstrual cyclicity in overweight women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(7):2670-8. Available from: https://doi.org/10.1210/jc.2008-0115
- Elkind-Hirsch KE, Chappell N, Seidemann E, Storment J, Bellanger D. Exenatide, Dapagliflozin, or Phentermine/Topiramate Differentially Affect Metabolic Profiles in Polycystic Ovary Syndrome. J Clin Endocrinol Metab. 2021;106(10):3019-33. Available from: https://doi.org/10.1210/clinem/dgab408
- Liu X, Zhang Y, Zheng SY, Lin R, Xie YJ, Chen H, et al. Efficacy of exenatide on weight loss, metabolic parameters and pregnancy in overweight/obese polycystic ovary syndrome. Clin Endocrinol (Oxf). 2017;87(6):767-74. Available from: https://doi.org/10.1111/cen.13454
- Ma RL, Deng Y, Wang YF, Zhu SY, Ding XS, Sun AJ. Short-term combined treatment with exenatide and metformin for overweight/obese women with polycystic ovary syndrome. Chin Med J (Engl). 2021;134(23):2882-9. Available from: https://doi.org/10.1097/cm9.0000000000001712
- Tao T, Zhang Y, Zhu YC, Fu JR, Wang YY, Cai J, et al. Exenatide, Metformin, or Both for Prediabetes in PCOS: A Randomized, Open-label, Parallel-group Controlled Study. J Clin Endocrinol Metab. 2021;106(3):e1420-e32. Available from: https://doi.org/10.1210/clinem/dgaa692
- Elkind-Hirsch KE, Chappell N, Shaler D, Storment J, Bellanger D. Liraglutide 3 mg on weight, body composition, and hormonal and metabolic parameters in women with obesity and polycystic ovary syndrome: a randomized placebo-controlled-phase 3 study. Fertil Steril. 2022;118(2):371-81. Available from: https://doi.org/10.1016/j.fertnstert.2022.04.027
- Frøssing S, Nylander M, Chabanova E, Frystyk J, Holst JJ, Kistorp C, et al. Effect of liraglutide on ectopic fat in polycystic ovary syndrome: A randomized clinical trial. Diabetes Obes Metab. 2018;20(1):215-8. Available from: https://doi.org/10.1111/dom.13053
- Liao CC, Chien CH, Yu TS, Li JM. Integrated Chinese and Western Medicine for Coronary Heart Disease Prevention in Polycystic Ovary Syndrome: A 19-Year Nationwide Cohort Study. J Multidiscip Healthc. 2023;16:3959-73. Available from: https://doi.org/10.2147/jmdh.S442816
- Xing C, Zhao H, Zhang J, He B. Effect of metformin versus metformin plus liraglutide on gonadal and metabolic profiles in overweight patients with polycystic ovary syndrome. Front Endocrinol (Lausanne). 2022;13:945609. Available from: https://doi.org/10.3389/fendo.2022.945609
- Wen Q, Fang S, Liang Y, Tian Y, Chen Y, Yuan J, et al. Short-term effect of beinaglutide combined with metformin versus metformin alone on weight loss and metabolic profiles in obese patients with polycystic ovary syndrome: a pilot randomized trial. Front Endocrinol (Lausanne). 2023;14:1156521. Available from: https://doi.org/10.3389/fendo.2023.1156521
- Zhang Y, Qu Z, Lu T, Shao X, Cai M, Dilimulati D, et al. Effects of a Dulaglutide plus Calorie-Restricted Diet versus a Calorie-Restricted Diet on Visceral Fat and Metabolic Profiles in Women with Polycystic Ovary Syndrome: A Randomized Controlled Trial. Nutrients. 2023;15(3). Available from: https://doi.org/10.3390/nu15030556
- Frøssing S, Nylander M, Kistorp C, Skouby SO, Faber J. Effect of liraglutide on atrial natriuretic peptide, adrenomedullin, and copeptin in PCOS. Endocr Connect. 2018;7(1):115-23. Available from: https://doi.org/10.1530/ec-17-0327
- Nylander M, Frøssing S, Clausen HV, Kistorp C, Faber J, Skouby SO. Effects of liraglutide on ovarian dysfunction in polycystic ovary syndrome: a randomized clinical trial. Reprod Biomed Online. 2017;35(1):121-7. Available from: https://doi.org/10.1016/j.rbmo.2017.03.023
- Nylander M, Frøssing S, Kistorp C, Faber J, Skouby SO. Liraglutide in polycystic ovary syndrome: a randomized trial, investigating effects on thrombogenic potential. Endocr Connect. 2017;6(2):89-99. Available from: https://doi.org/10.1530/ec-16-0113
- Li R, Mai T, Zheng S, Zhang Y. Effect of metformin and exenatide on pregnancy rate and pregnancy outcomes in overweight or obese infertility PCOS women: long-term follow-up of an RCT. Arch Gynecol Obstet. 2022;306(5):1711-21. Available from: https://doi.org/10.1007/s00404-022-06700-3
- Zheng S, Zhang Y, Long T, Lu J, Liu X, Yan J, et al. Short term monotherapy with exenatide is superior to metformin in weight loss, improving insulin resistance and inflammation in Chinese overweight/obese PCOS women. Obesity Medicine. 2017;7. Available from: https://doi.org/10.1016/j.obmed.2017.06.003
- Gan J, Chen J, Ma RL, Deng Y, Ding XS, Zhu SY, et al. Metagenomics study on taxonomic and functional change of gut microbiota in patients with obesity with PCOS treated with exenatide combination with metformin or metformin alone. Gynecol Endocrinol. 2023;39(1):2219342. Available from: https://doi.org/10.1080/09513590.2023.2219342
- El Maghraby HA, Nafee T, Guiziry D, Elnashar A. Randomized controlled trial of the effects of metformin versus combined oral contraceptives in adolescent PCOS women through a 24month follow up period. Middle East Fertility Society Journal. 2015;20(3):131-7. Available from: https://doi.org/10.1016/j.mefs.2014.10.003
- Bhattacharya SM, Jha A. Comparative study of the therapeutic effects of oral contraceptive pills containing desogestrel, cyproterone acetate, and drospirenone in patients with polycystic ovary syndrome. Fertil Steril. 2012;98(4):1053-9. Available from: https://doi.org/10.1016/j.fertnstert.2012.06.035
- Fonseka S, Wijeyaratne CN, Gawarammana IB, Kalupahana NS, Rosairo S, Ratnatunga N, et al. Effectiveness of Low-dose Ethinylestradiol/Cyproterone Acetate and Ethinylestradiol/Desogestrel with and without Metformin on Hirsutism in Polycystic Ovary Syndrome: A Randomized, Double-blind, Triple-dummy Study. J Clin Aesthet Dermatol. 2020;13(7):18-23.
- Kahraman K, Sükür YE, Atabekoğlu CS, Ateş C, Taşkın S, Cetinkaya SE, et al. Comparison of two oral contraceptive forms containing cyproterone acetate and drospirenone in the treatment of patients with polycystic ovary syndrome: a randomized clinical trial. Arch Gynecol Obstet. 2014;290(2):321-8. Available from: https://doi.org/10.1007/s00404-014-3217-5
- Mastorakos G, Koliopoulos C, Creatsas G. Androgen and lipid profiles in adolescents with polycystic ovary syndrome who were treated with two forms of combined oral contraceptives. Fertil Steril. 2002;77(5):919-27. Available from: https://doi.org/10.1016/s0015-0282(02)02993-x
- Mastorakos G, Koliopoulos C, Deligeoroglou E, Diamanti-Kandarakis E, Creatsas G. Effects of two forms of combined oral contraceptives on carbohydrate metabolism in adolescents with polycystic ovary syndrome. Fertil Steril. 2006;85(2):420-7. Available from: https://doi.org/10.1016/j.fertnstert.2005.07.1306
- Yildizhan R, Gokce AI, Yildizhan B, Cim N. Comparison of the effects of chlormadinone acetate versus drospirenone containing oral contraceptives on metabolic and hormonal parameters in women with PCOS for a period of two-year follow-up. Gynecol Endocrinol. 2015;31(5):396-400. Available from: https://doi.org/10.3109/09513590.2015.1006187
- Bhattacharya SM, Jha A, DasMukhopadhyay L. Comparison of two contraceptive pills containing drospirenone and 20 μg or 30 μg ethinyl estradiol for polycystic ovary syndrome. Int J Gynaecol Obstet. 2016;132(2):210-3. Available from: https://doi.org/10.1016/j.ijgo.2015.06.065
- Glintborg D, Altinok ML, Mumm H, Hermann AP, Ravn P, Andersen M. Body composition is improved during 12 months' treatment with metformin alone or combined with oral contraceptives compared with treatment with oral contraceptives in polycystic ovary syndrome. J Clin Endocrinol Metab. 2014;99(7):2584-91. Available from: https://doi.org/10.1210/jc.2014-1135
- Harborne L, Fleming R, Lyall H, Sattar N, Norman J. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88(9):4116-23. Available from: https://doi.org/10.1210/jc.2003-030424
- Palomba S, Falbo A, Russo T, Manguso F, Tolino A, Zullo F, et al. Insulin sensitivity after metformin suspension in normal-weight women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2007;92(8):3128-35. Available from: https://doi.org/10.1210/jc.2007-0441
- Alpañés M, Álvarez-Blasco F, Fernández-Durán E, Luque-Ramírez M, Escobar-Morreale HF. Combined oral contraceptives plus spironolactone compared with metformin in women with polycystic ovary syndrome: a one-year randomized clinical trial. Eur J Endocrinol. 2017;177(5):399-408. Available from: https://doi.org/10.1530/eje-17-0516
- de Zegher F, Díaz M, Villarroya J, Cairó M, López-Bermejo A, Villarroya F, et al. The relative deficit of GDF15 in adolescent girls with PCOS can be changed into an abundance that reduces liver fat. Scientific Reports. 2021;11(1). Available from: https://doi.org/10.1038/s41598-021-86317-9
- Díaz M, Gallego-Escuredo JM, López-Bermejo A, De Zegher F, Villarroya F, Ibáñez L. Low-Dose Spironolactone-Pioglitazone-Metformin Normalizes Circulating Fetuin-A Concentrations in Adolescent Girls with Polycystic Ovary Syndrome. International Journal of Endocrinology. 2018;2018. Available from: https://doi.org/10.1155/2018/4192940
- Ibáñez L, del Río L, Díaz M, Sebastiani G, Pozo ÓJ, López-Bermejo A, et al. Normalizing Ovulation Rate by Preferential Reduction of Hepato-Visceral Fat in Adolescent Girls With Polycystic Ovary Syndrome. Journal of Adolescent Health. 2017;61(4):446-53. Available from: https://doi.org/10.1016/j.jadohealth.2017.04.010
- Ibáñez L, Díaz M, García-Beltrán C, Malpique R, Garde E, López-Bermejo A, et al. Toward a Treatment Normalizing Ovulation Rate in Adolescent Girls With Polycystic Ovary Syndrome. J Endocr Soc. 2020;4(5):bvaa032. Available from: https://doi.org/10.1210/jendso/bvaa032
- Malpique R, Sánchez-Infantes D, Garcia-Beltran C, Taxeras SD, López-Bermejo A, de Zegher F, et al. Towards a circulating marker of hepato-visceral fat excess: S100A4 in adolescent girls with polycystic ovary syndrome — Evidence from randomized clinical trials. Pediatric Obesity. 2019;14(5). Available from: https://doi.org/10.1111/ijpo.12500
- Moretti C, Guccione L, Di Giacinto P, Simonelli I, Exacoustos C, Toscano V, et al. Combined Oral Contraception and Bicalutamide in Polycystic Ovary Syndrome and Severe Hirsutism: A Double-Blind Randomized Controlled Trial. J Clin Endocrinol Metab. 2018;103(3):824-38. Available from: https://doi.org/10.1210/jc.2017-01186
- Spritzer PM, Lisboa KO, Mattiello S, Lhullier F. Spironolactone as a single agent for long-term therapy of hirsute patients. Clinical Endocrinology. 2000;52(5):587-94. Available from: https://doi.org/10.1046/j.1365-2265.2000.00982.x
- Kumarendran B, O'Reilly MW, Subramanian A, Šumilo D, Toulis K, Gokhale KM, et al. Polycystic Ovary Syndrome, Combined Oral Contraceptives, and the Risk of Dysglycemia: A Population-Based Cohort Study With a Nested Pharmacoepidemiological Case-Control Study. Diabetes Care. 2021;44(12):2758-66. Available from: https://doi.org/10.2337/dc21-0437
- Chen SF, Yang YC, Hsu CY, Shen YC. Risk of bipolar disorder in patients with polycystic ovary syndrome: A nationwide population-based cohort study. J Affect Disord. 2020;263:458-62. Available from: https://doi.org/10.1016/j.jad.2019.12.007
- Chen SF, Yang YC, Hsu CY, Shen YC. Risk of schizophrenia in patients with polycystic ovary syndrome: a nationwide population-based cohort study from Taiwan. J Psychosom Obstet Gynaecol. 2021;42(4):272-8. Available from: https://doi.org/10.1080/0167482x.2020.1735342
- Park B, Lee H, Park S, Lee ES, Lee JJ, Lee Y, et al. Trend changes and factor analysis of endometrial hyperplasia in patients with polycystic ovarian syndrome based on the Korean National Health Insurance Database. BMC Womens Health. 2022;22(1):439. Available from: https://doi.org/10.1186/s12905-022-02015-2
- Läkemedelsverket. Läkemedelsverkets syn på off-label-förskrivning. Stockholm: Läkemedelsverket; 2020. [updated 24 Sep 2020; accessed 18 Jun]. Available from: https://www.lakemedelsverket.se/sv/behandling-och-forskrivning/forskrivning/skriva-recept/lakemedelsverkets-syn-pa-off-label-forskrivning#hmainbody1
- Fortsatt vädjan om att förskriva GLP-1-receptoragonister endast inom godkänd indikation [press release]. Stockholm: Läkemedelsverket, 2025.
- Arthurson V, Sundstrom A, Ljung R. Förskriv GLP-1-receptoragonister endast på godkänd indikation. Läkartidningen. 2023 11 Dec.
- Apridonidze T, Essah PA, Iuorno MJ, Nestler JE. Prevalence and characteristics of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2005;90(4):1929-35. Available from: https://doi.org/10.1210/jc.2004-1045
- Coffey S, Bano G, Mason HD. Health-related quality of life in women with polycystic ovary syndrome: a comparison with the general population using the Polycystic Ovary Syndrome Questionnaire (PCOSQ) and the Short Form-36 (SF-36). Gynecol Endocrinol. 2006;22(2):80-6. Available from: https://doi.org/10.1080/09513590600604541
- Socialstyrelsen. Nationella riktlinjer vid vård för obesitas - Prioriteringsstöd till beslutsfattare och chefer 2023. Stockholm: Socialstyrelsen; 2023. 2023-4-8460. [updated 27 Mar 2023]. Available from: https://www.socialstyrelsen.se/publikationer/nationella-riktlinjer-for-vard-vid-obesitas--prioriteringsstod-till-beslutsfattare-och-chefer-2023-2023-4-8460/
- Endokrinologi. SFOG. [updated Apr 4 2025; accessed Jul 9 2025]. Available from: https://www.sfog.se/kunskap/endokrinologi/
- God och nära vård Stockholm: Socialstyrelsen. [updated Apr 3 2025; accessed Jul 9 2025]. Available from: https://www.socialstyrelsen.se/kunskapsstod-och-regler/omraden/god-och-nara-vard/
- Lau GM, Elghobashy M, Thanki M, Ibegbulam S, Latthe P, Gillett CDT, et al. A systematic review of lived experiences of people with polycystic ovary syndrome highlights the need for holistic care and co-creation of educational resources. Frontiers in Endocrinology. 2022;Volume 13 - 2022. Available from: https://doi.org/10.3389/fendo.2022.1064937
- Mottagning Medicinsk hårborttagning. Stockholm: Karolinska Universitetssjukhuset (KI). [updated Jun 18 2025; accessed Jul 8 2025]. Available from: https://www.karolinska.se/vard/tema/tema-inflammation-och-aldrande/endokrinologi/sidmall-mottagning-avdelning/
- HEE L, KETTNER LO, VEJTORP M. Continuous use of oral contraceptives: an overview of effects and side-effects. Acta Obstetricia et Gynecologica Scandinavica. 2013;92(2):125-36. Available from: https://doi.org/10.1111/aogs.12036
- Riktlinjer för behandling med hormonella preventivmetoder. Stockholm: Janusinfo Region Stockholm. [updated Nov 5 2024; accessed Jul 9 2025]. Available from: https://janusinfo.se/behandling/expertgruppsutlatanden/kvinnosjukdomarochforlossning/kvinnosjukdomarochforlossning/riktlinjerforbehandlingmedhormonellapreventivmetoder.5.6081a39c160e9b387319df.html
- GRADE working group. The Grading of Recommendations Assessment, Development and Evaluation (GRADE). [accessed Jul 8 2025]. Available from: https://www.gradeworkinggroup.org/
- Oberg E, Gidlof S, Jakson I, Mitsell M, Tollet Egnell P, Hirschberg AL. Improved menstrual function in obese women with polycystic ovary syndrome after behavioural modification intervention-A randomized controlled trial. Clin Endocrinol (Oxf). 2019;90(3):468-78. Available from: https://doi.org/10.1111/cen.13919
- Liu Y, Ruan B, Jiang H, Le S, Liu Y, Ao X, et al. The Weight-loss Effect of GLP-1RAs Glucagon-Like Peptide-1 Receptor Agonists in Non-diabetic Individuals with Overweight or Obesity: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Am J Clin Nutr. 2023;118(3):614-26. Available from: https://doi.org/10.1016/j.ajcnut.2023.04.017
- Cesta CE, Mansson M, Palm C, Lichtenstein P, Iliadou AN, Landén M. Polycystic ovary syndrome and psychiatric disorders: Co-morbidity and heritability in a nationwide Swedish cohort. Psychoneuroendocrinology. 2016;73:196-203. Available from: https://doi.org/10.1016/j.psyneuen.2016.08.005
- Norwegian Register for Scientific Journals Series and Publishers. The Norwegian Directorate for Higher Education and Skills. Bergen. [updated March 6 2024; accessed April 23 2025]. Available from: https://kanalregister.hkdir.no/en/informasjonsartikler/search-in-the-register
Scientific article
Forslund M, Wändell P, Forsberg L, Österberg M, Dagerhamn J, Wernersson E, et al. GLP-1 receptor agonist treatment in women with polycystic ovary syndrome-a systematic review and meta-analysis. Eur J Endocrinol. 2026;194(3):25-39. Available from: https://doi.org/10.1093/ejendo/lvag033

 Swedish Agency for Health Technology Assessment and Assessment of Social Services
Swedish Agency for Health Technology Assessment and Assessment of Social Services
 Share on Facebook
Share on Facebook
 Share on LinkedIn
Share on LinkedIn
 Share via Email
Share via Email