This publication was published more than 5 years ago. The state of knowledge may have changed.
Dabigatran to Prevent Stroke in Patients With Atrial Fibrillation
Summary and conclusions
Atrial fibrillation is the most common type of cardiac arrhythmia. Atrial fibrillation increases the risk of forming blood clots, clots that the circulatory system carries through the body. These clots may cause vascular events such as stroke. To prevent stroke, doctors use drugs that reduce the blood’s ability to coagulate (anticoagulants) when treating many patients who present with atrial fibrillation and at least one additional stroke risk factor. Warfarin is commonly used, but the drug must be carefully managed since it carries a risk for serious bleeding. Dabigatran is a new type of anticoagulant with a different mechanism of action. Dabigatran has been compared to warfarin therapy, and a recently published analysis of the study compares results from four categories of centres where warfarin therapy has been managed with greater or lesser success. Since previous research shows that warfarin therapy is usually managed very well in Sweden, this SBU Alert report is based mainly on study results from the centres that manage warfarin therapy most successfully. Hence, the conclusions of this report are based on the assumption that the quality of warfarin therapy in Sweden will continue to be very well managed. Consequently, follow-up and monitoring of the quality of warfarin therapy is of major importance.
SBU’s appraisal of the evidence
- An overall appraisal of the medical benefits and risks for the patient group as a whole does not show dabigatran to be superior to warfarin in treating patients with atrial fibrillation and a higher risk of stroke. Assuming that warfarin therapy is well managed, which is the norm in Sweden, dabigatran and warfarin show no substantial differences in risk, either for stroke and other types of blood clots, or for serious haemorrhaging generally or death regardless of cause. The specific risk for cerebral haemorrhage appears, however, to be lower with dabigatran than with warfarin even when warfarin therapy is well managed, but this must be viewed against the potential risks of dabigatran.
- Conclusions from clinical studies are based on comparisons at the group level. Despite the conclusion presented under the first point, there are presumably individuals for whom dabigatran yields a better balance between risks and benefits than warfarin does, eg, patients who have tried warfarin and for whom the dose could not be successfully established.
- For patients where warfarin therapy was deemed inappropriate (contraindicated) from the outset, no evidence is available to appraise the benefits and risks of dabigatran.
- Given the current price of dabigatran, and based on a comparison of centres with high-quality management of warfarin, warfarin therapy is the most cost-effective option. Since the health economic analysis is sensitive to costs associated with visits for specimen taking, there may be individuals for whom dabigatran is, for various reasons, more cost-effective than warfarin therapy.
Technology and target group
Atrial fibrillation is the most common type of cardiac arrhythmia. The condition is estimated to affect 100 000 people in Sweden, and it is more common at higher ages. Atrial fibrillation is associated with an annual risk for stroke of 3 to 5 percent. Examples of other factors that contribute to further increasing the risk of atrial fibrillation are smoking, hypertension, and diabetes along with other cardiac disorders such as congestive heart failure, angina, and cardiomyopathy.
Vitamin K antagonists are often prescribed to reduce the ability of blood to coagulate and thereby prevent blood clots in individuals with atrial fibrillation. Currently, warfarin is most commonly used, and patients with atrial fibrillation comprise the largest patient group using the drug. The sensitivity of warfarin varies among individuals, and in the same individual over time, and is influenced, eg, by diet and other drugs.
Warfarin therapy carries a risk for severe and life-threatening haemorrhage. Treatment involves finding a balance between benefits (protection against blood clots and stroke) and risks (side effects such as severe, occasionally life-threatening, haemorrhage). Hence, it is important to appropriately inhibit the blood’s ability to coagulate. Consequently, warfarin therapy requires that patients are checked regularly and that dosage is adjusted to maximise protective effects while minimising potential risks for side effects.
Warfarin therapy is managed by analysing prothrombin complex (PC), a service provided in Sweden by special anticoagulation clinics (AC clinics) or primary care. Not every patient with atrial fibrillation and an elevated stroke risk can be treated with warfarin due to the risks or difficulties in managing this complicated treatment.
Dabigatran is a new type of anticoagulant medication with a different mechanism of action; it is a direct thrombin inhibitor.
The target group for dabigatran treatment includes individuals with atrial fibrillation and an elevated risk of stroke, but who do not have heart valve problems. The study in this report included only patients appropriate for warfarin therapy.
Primary questions
- In weighing benefits and risks, are there any overall differences between dabigatran treatment and warfarin therapy?
- The assessment also aims to compare the costs and cost-effectiveness of dabigatran treatment and warfarin therapy.
Patient benefit
Assuming that the management of warfarin therapy is of good quality, corresponding to standard practice in Swedish health care, the following apply:
- Moderately strong scientific evidence shows no clinically relevant differences in the risk of stroke or systemic embolisation between patients treated with dabigatran and patients treated with warfarin (
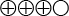 ).
). - Moderately strong scientific evidence shows no clinically relevant differences in the risk of severe haemorrhage between patients treated with dabigitran and patients treated with warfarin (
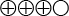 ).
). - Moderately strong scientific evidence shows a lower risk of cerebral haemorrhage in patients treated with dabigatran compared to patients treated with warfarin (
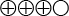 ).
). - Moderately strong scientific evidence shows no apparent difference in the risk of death, regardless of cause, between patients treated with dabigatran and patients treated with warfarin (
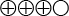 ).
).
Ethical aspects
The report raises a series of potential conflicts of interest and values among patients, taxpayers, corporations, and professions.
Current evidence reveals no general superiority of one treatment over the other at the group level and shows that warfarin therapy is generally the most cost-effective option. However, there may be individual patients for whom warfarin therapy is less appropriate, or completely inappropriate, due to medical reasons. Dabigatran could be a treatment option in such cases. Furthermore, certain individuals may experience comfort- or autonomy-related advantages with dabigatran.
Economic aspects
Since a comparison of centres providing high-quality management of warfarin showed that neither treatment is generally medically superior to the other, we conducted a cost minimisation analysis. Based on the current price of dabigatran, the analysis shows that warfarin therapy is the most cost-effective option. Since the health economic analysis is sensitive to costs associated with visits for specimen taking, dabigatran treatment could be more cost-effective than warfarin therapy in certain individual cases.
Four levels are used in grading the strength of the scientific evidence on which conclusions are based:
Strong scientific evidence (![]() ). Based on high or medium quality studies with no factors that weaken the overall assessment.
). Based on high or medium quality studies with no factors that weaken the overall assessment.
Moderately strong scientific evidence (![]() ). Based on high or medium quality studies with isolated factors that weaken the overall assessment.
). Based on high or medium quality studies with isolated factors that weaken the overall assessment.
Limited scientific evidence (![]() ). Based on high or medium quality studies having factors that weaken the overall assessment.
). Based on high or medium quality studies having factors that weaken the overall assessment.
Insufficient scientific evidence (![]() ). Scientific evidence is deemed insufficient when scientific findings are absent, the quality of available studies is low, or studies of similar quality present conflicting findings.
). Scientific evidence is deemed insufficient when scientific findings are absent, the quality of available studies is low, or studies of similar quality present conflicting findings.
References
- Hart RG, Pearce LA, Aguilar MI. Meta-analysis: Antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med 2007;146(12):857-67.
- Fuster V, Rydén LE, Cannom DS, Crijns HJ, Curtis AB, Ellenbogen KA, et al. ACC/AHA/ESC 2006 Guidelines for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation): Developed in Collaboration With the European Heart Rhythm Association and the Heart Rhythm Society. Circulation 2006;114(7):e257-354.
- Fryckstedt J, Asker-Hagelberg C. [Drug-related problems common in the emergency department of internal medicine. The cause of admission in almost every third patient according to quality follow-up]. Läkartidningen 2008;105(12-13):894-8.
- Läkemedelsverket. Rapporterade biverkningar 2008. http://www.lakemedelsverket.se/Alla-nyheter/NYHETER-2009/Rapporterade-biverkningar-2008/
- Hillarp A, Egberg N, Nordin G, Stigendal L, Fagerberg I, Lindahl TL. Local INR calibration of the Owren type prothrombin assay greatly improves the intra- and interlaboratory variation. A three-year follow-up from the Swedish national external quality assessment scheme. Thromb Haemost 2004;91(2):300-7.
- Rosendaal FR, Cannegieter SC, van der Meer FJ, Briët E. A method to determine the optimal intensity of oral anticoagulant therapy. Thromb Haemost 1993;69(3):236-9.
- Boudin F, Nie JY, Bartlett JC, Grad R, Pluye P, Dawes M. Combining classifiers for robust PICO element detection. BMC Med Inform Decis Mak 2010;10:29.
- Holbrook AM, Pereira JA, Labiris R, McDonald H, Douketis JD, Crowther M, et al. Systematic overview of warfarin and its drug and food interactions. Arch Intern Med 2005;165(10):1095-106.
- Rosenqvist M, Själander A, January 2011, personal communication.
- Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009;361(12):1139-51.
- Connolly SJ, Ezekowitz MD, Yusuf S, Reilly PA, Wallentin L. Newly identified events in the RE-LY trial. N Engl J Med 2010;363(19):1875-6.
- Wallentin L, Yusuf S, Ezekowitz MD, Alings M, Flather M, Franzosi MG, et al. Efficacy and safety of dabigatran compared with warfarin at different levels of international normalised ratio control for stroke prevention in atrial fibrillation: an analysis of the RE-LY trial. Lancet 2010;376(9745):975-83.
- Earl MA, Mauro VF. The Clinical Impact of Administering Warfarin to Post-Myocardial Infarction Patients. Journal of Pharmacy Practice 2004;17:266-9.
- Riks-Stroke, Årsrapporter, http://www.riks-stroke.org/index.php?content=analyser
- SBU. Självtestning och egenvård vid användning av blodproppsförebyggande läkemedel. En systematisk litteraturöversikt. Stockholm: Statens beredning för medicinsk utvärdering (SBU); Alert-rapport nr 2007-05. ISSN 1652-7151. http://www.sbu.se
- Davidson T, Levin L-Å. Kostnaden för förmaksflimmer i Östergötland. Linköpings universitet; CMT Rapport 2006:5. ISSN 0283-1228.
- De nya läkemedelsförmånerna. Regeringens propostition 2001/02:63 (p 44). http://www.regeringen.se
- Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336(7650):924-6.
 Swedish Agency for Health Technology Assessment and Assessment of Social Services
Swedish Agency for Health Technology Assessment and Assessment of Social Services




